PROFID was featured prominently in two separate sessions at the European Heart Rhythm Association congress – EHRA 2022 in Copenhagen (3 April – 5 April 2022). The latest results of the PROFID analysis were presented to leading scientists and key opinion leaders in cardiology from all over the world.
It was a great occasion for us to showcase our latest findings which you can watch here:
Session: Late Breaking Clinical Trials I – devices and prevention sudden cardiac death
Title: The updated PROFID clinical prediction model.
Speaker: Doctor N. Dagres (Leipzig, DE)
Session: The EHRA EU research projects
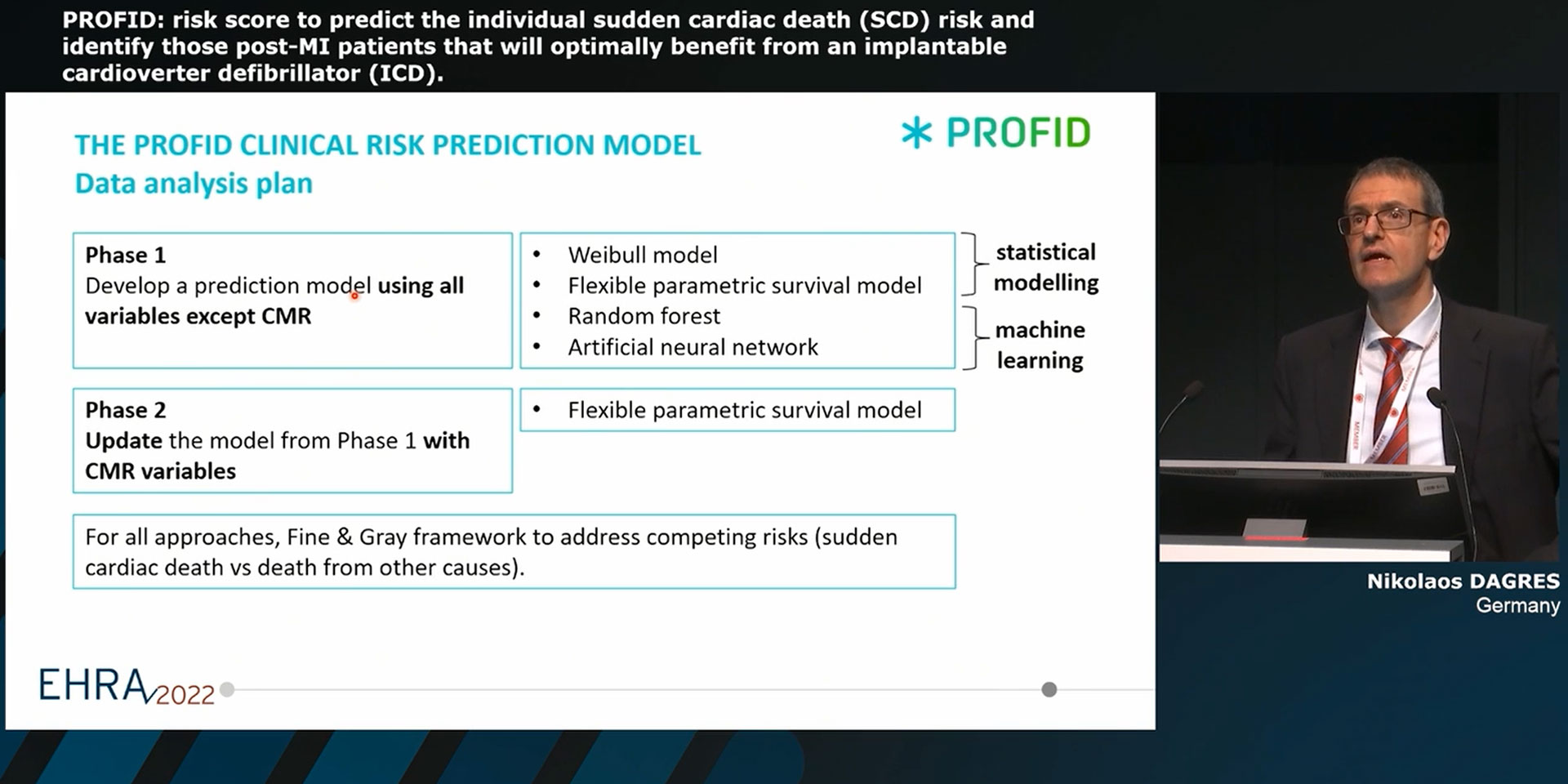
Title: PROFID: risk score to predict the individual sudden cardiac death (SCD) risk and identify those post-MI patients that will optimally benefit from an implantable cardioverter defibrillator (ICD).
Speaker: Doctor N. Dagres (Leipzig, DE)
This presentation was accompanied by a press release distributed by the ESC and published on their website:
PROFID project status
The ambition of PROFID is to introduce a disruptive innovation in the prevention of SCD in post-myocardial infarction (MI) patients by introducing – for the first time – a personalised prevention approach.
Phase one of the PROFID analysis in 19 datasets, from Europe, Israel and the US, was based on a collection of existing highly phenotyped data with the largest number of post-MI patients (~225,000 observations). The datasets, accessed from 13 partners, represent a wide variety of data sources such as national registries, institutional research databases, electronic health records and clinical trials. Four models were developed using traditional analytical methods or artificial intelligence techniques (Weibull model, flexible parametric survival model, random forest, deep neural network). LVEF was found to be a poor predictor of SCD. Consideration of the large variety of additional variables (demographics, clinical parameters, medication, ECG, biomarkers, echocardiograph etc.) also did not improve predictive performance.
In phase two of the PROFID analysis, we updated the prediction model with inclusion of cardiac magnetic resonance (CMR) variables. Previous studies have indicated a predictive value of fibrosis detection by CMR for the assessment of SCD risk. The visualization of the peri-infarct zone may be more specific for SCD compared with the other risk stratification methods, since the majority of the arrhythmias leading to SCD originate in the border zone between the necrotic and the healthy myocardial tissue. However, there is significant heterogeneity in the definition of scar and grayzone in the literature that limits the interpretation of study findings. In order to unmask the true potential of this data for SCD prediction, we reanalysed 7 datasets from Europe and the United States comprising patients with previous MI (regardless of LVEF), or ischemic cardiomyopathy with LVEF<50%, in whom CMR had been performed at least 40 days after MI. With ~2.100 patients, we are presenting the largest cohort of CMR data analysed for predicting the risk of SCD. An extensive re-evaluation of the CMR studies was performed in order to achieve consistent CMR measurements of scar size and grayzone size across all 7 datasets, to reduce measurement heterogeneity and compare different previously applied thresholds for core scar and grayzone quantification, for instance standard deviation (2SD, 3SD, 5SD), and full-width-half-max methods. These variables were then added into the model via systematic internal-external cross-validation using a flexible parametric model fit under a cause-specific competing risk framework. The results indicate a potentially significant role of CMR for risk stratification for SCD after infarction.
In the next step we will continue our reanalysis further updating the model with different combinations of the CMR measurements in order to inform the upcoming randomised multi-centre clinical trials, PROFID-Reduced and PROFID-Preserved.